Abogen, in collaboration with the National University of Malaysia (UKM), has successfully obtained approval for the joint project under the “Intergovernmental International Science and Technology Innovation Cooperation” special program of the National Key R&D Program of China.
The project, titled “Development of a Novel Nasal Spray mRNA Tuberculosis (TB) Vaccine: Mcosal-Anchored Adjuvant Nanoparticle Delivery Technology” (Project No: 2025YFE0115700), has been officially approved by the Ministry of Science and Technology (MOST) of China and the Ministry of Science, Technology and Innovation (MOSTI) of Malaysia as a bilateral intergovernmental initiative.
As a cornerstone of China’s National Key R&D Program, the “Intergovernmental International Science, Technology and Innovation (STI) Cooperation” initiative aims to pool global expertise to tackle frontier scientific challenges and key core technologies. The program maintains an exceptionally rigorous selection process, requiring projects to demonstrate world-class innovation and promising application prospects, while fostering substantive, complementary partnerships with leading international teams. Consequently, the annual call for proposals attracts top-tier research institutes, universities, and industry leaders nationwide. Following multiple rounds of stringent review, only a select few emerge successful — a testament to the high standards and immense strategic value of this prestigious national program.
Tuberculosis (TB) remains one of the world’s most significant infectious disease threats. According to the World Health Organization’s (WHO) Global Tuberculosis Report 2025, global TB diagnoses reached 8.3 million cases in 2024, hitting a new historical peak in surveillance. With 1.23 million deaths, TB has surpassed HIV/AIDS and COVID-19 to become the leading cause of death from a single infectious agent globally. Despite this crisis, no TB vaccine for adults has yet been approved for market, making the development of next-generation preventive measures an urgent priority for international public health.
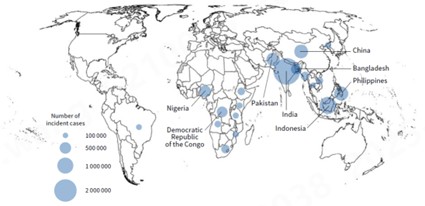
Figure: Countries with an estimated tuberculosis incidence of 100,000 cases or more in 2024
mRNA vaccines are uniquely effective for tuberculosis prevention, as they trigger both specific humoral immunity and high-level cellular immune responses. This project focuses on the cutting edge of mRNA-LNP delivery, aiming to develop a novel TB vaccine utilizing an innovative Lipid Nanoparticle (LNP) system to achieve highly efficient and safe nasal mucosal immunity. Intranasal administration is particularly critical because it directly targets the respiratory mucosa—the first line of defense against Mycobacterium tuberculosis. By preventing infection at the source and blocking transmission, this approach significantly strengthens the collective immunity barrier.
This international collaboration represents a powerful synergy of complementary strengths. Universiti Kebangsaan Malaysia (UKM) is one of Malaysia’s premier research universities. The team led by Dr. Fazren Azmi is at the forefront of vaccine delivery research, having developed a specialized mucosal-anchored adjuvant lipid nanoparticle (LNP) platform. Specifically engineered for intranasal administration, this platform triggers robust mucosal immunity. Their MARE technology features mucosal anchoring and receptor-engaging capabilities, functions as a TLR5 agonist, and exhibits excellent biocompatibility, promising to significantly enhance delivery efficiency.
Abogen brings to the partnership a proprietary library of ionizable lipids, clinically validated through trials involving over 30,000 participants—making it one of the few mRNA companies globally to achieve such international clinical verification. Currently, the company’s pipeline includes lyophilized mRNA vaccines for Shingles and Respiratory Syncytial Virus (RSV), both of which have advanced to Phase II clinical trials. Abogen also possesses a solid research foundation in TB mRNA vaccines, having completed antigen screening, lyophilization process development, and guinea pig challenge protection studies in collaboration with authoritative domestic institutions.
By combining cutting-edge mRNA technology with extensive TB research expertise, both parties are uniquely positioned to jointly pioneer a new generation of intranasal mRNA tuberculosis vaccines.
The socio-economic impact of a dedicated adult tuberculosis vaccine is immense. In its report, An Investment Case for New Tuberculosis Vaccines, the World Health Organization (WHO) identifies the development of an effective vaccine for adolescents and adults as the decisive factor in ending the TB epidemic.
The report projects that global vaccine demand for these age groups will reach between 4.64 billion and 5.18 billion doses from 2025 to 2050. The introduction of a vaccine with just 50% efficacy could avert an estimated 4.6 million to 8.5 million TB-related deaths during this period. From a socio-economic perspective, such an achievement is expected to generate $372 billion in incremental net monetary benefits and drive approximately $1.6 trillion (range: 0.8–3.0 trillion) in global GDP growth between 2025 and 2080.
As a cornerstone of China-Malaysia intergovernmental research collaboration, this project serves as a model for high-level scientific cooperation in active response to the Belt and Road Initiative (BRI). It represents a concrete realization of the China-Malaysia Community with a Shared Future through the lens of scientific innovation.
The partnership holds profound significance for promoting knowledge sharing, deepening diplomatic trust, and enhancing regional public health governance. By transforming cutting-edge mRNA technology into a truly accessible “global public good,” this initiative safeguards public well-being and provides a powerful new impetus for the high-quality development of a Global Community of Health for All.